CARTILAGE
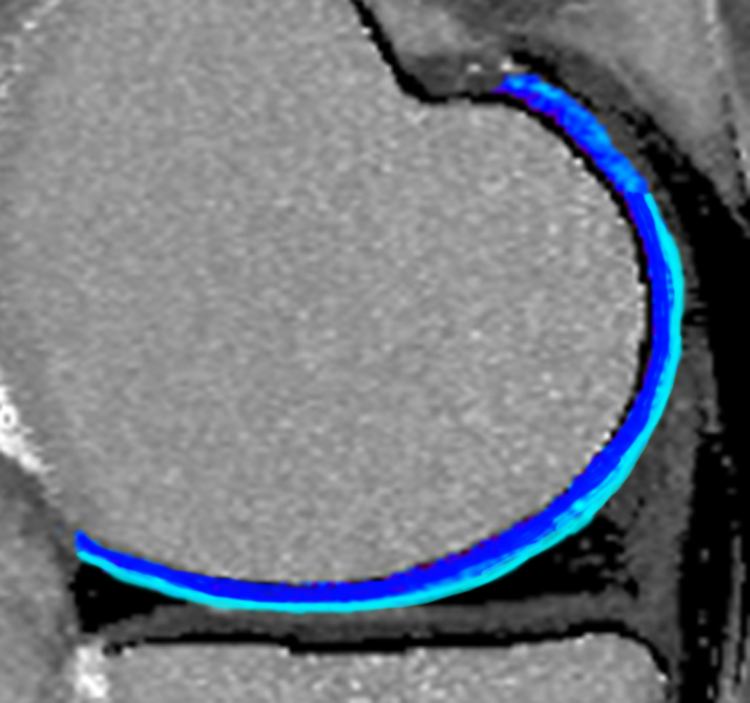
Early imaging of cartilage injury is an important step in joint preservation surgery and the development of new therapeutic strategies to restore and regenerate articular cartilage. Radiographs are the gold standard in osteoarthritis imaging, but indirectly image cartilage. New techniques in quantitative MRI have the ability to directly image the molecular structure of cartilage and predict arthritis development. Our group has engaged in clinical studies working with the National Institute of Health’s Osteoarthritis Initiative to develop classification software to identify imaging biomarkers in recognizing the development of and early progression of arthritis. This work has resulted in a pending United States patent.
Important software developed by our group includes ImageK – A program useful for multimodality rigid registration of three-dimensional MRI images. This is an important pre-processing step in using multiple qMRI image sequences in knee articular cartilage analysis. The software is freely available here.
PUBLICATIONS
- Zhong H, Miller DJ, Urish KL. (2016) T2 Map Signal Variation Predicts Symptomatic Osteoarthritis Progression: Data from the Osteoarthritis Initiative. Skeletal Radiology. 45(7) 909-913. PubMed PMID: 26992910.
- Urish KL, Keffalas MG, Durkin J, Miller DJ, Chu CR, Mosher, TJ. (2015) T2 texture index of cartilage can predict early symptomatic OA progression: Data from the Osteoarthritis Initiative. Osteoarthritis and Cartilage. 21(10), 1550-1557. PubMed PMID: 23774471.
- Urish KL, Williams A, Durkin J, Chu, CR, OAI Investigators Group. (2013) Registration of magnetic resonance image series for knee articular cartilage analysis: Data from the Osteoarthritis Initiative. Cartilage, 4(1) 20-27. PubMed PMID: 23997865.
- Durkin J, Miller DJ, Urish KL. (2011) Creating a 2D active shape model using itk::ImagePCAShapeModelEstimator. Insight Journal, //hdl.handle.net/10380/3282.

STEM CELLS
Dr. Urish has extensive experience on the role of oxidative stress in the regenerative capacity of adult donor-derived stem cells for muscle, cartilage, bone, and cardiac tissue regeneration. Our group has demonstrated that stem cells have an increased oxidative stress capacity. More importantly, this work demonstrated that the ability to handle oxidative stress directly effects the cell population’s differentiation and regenerative capacity. This work has demonstrated in vitro the possible efficacy of using anti-oxidant compounds and free radical scavengers in the development of stem cell therapies.
PUBLICATIONS
- Urish KL, Vella J, Okada M, Deasy BM, Tobita K, Keller BB, Cao B, Piganellie JD, Huard J. (2009) Antioxidant levels represent a major determinant in the regenerative capacity of muscle stem cells. Molecular Biology of the Cell, 20(1), 509-520. PubMed PMID: 19005220
- Deasy BM, Lu A, Tebbets JC, Feduska JM, Schugar RC, Pollett JB, Sun B, Urish KL, Gharaibeh BM, Cao B, Rubin RT, Huard J. (2007) A role for cell sex in stem cell mediated skeletal muscle regeneration: female cells have higher muscle regeneration efficiency. Journal of Cell Biology, 177(1), 73-86. PubMed PMID: 17420291.
- Urish KL, August J, Huard J. (2005) Unsupervised segmentation for myofiber counting in immunofluorescent microscopy images. Insight Journal, //hdl.handle.net/1926/48.
- Oshima H, Payne TR, Urish KL, Sakai T, Ling Y, Gharaibeh B, Tobita K, Keller BB, Cummins JH, Huard J. (2005) Differential myocardial infarct repair with muscle stem cells compared to myoblasts. Molecular Therapy, 12(6), 1130-1141. PubMed PMID: 16125468.
